2X weekly dosing can help align factor levels with the times you are physically active1
Talk to your doctor about which infusion days and dosage may be recommended for you. Your doctor will determine your dosage based on your age, weight, and bleeding pattern.

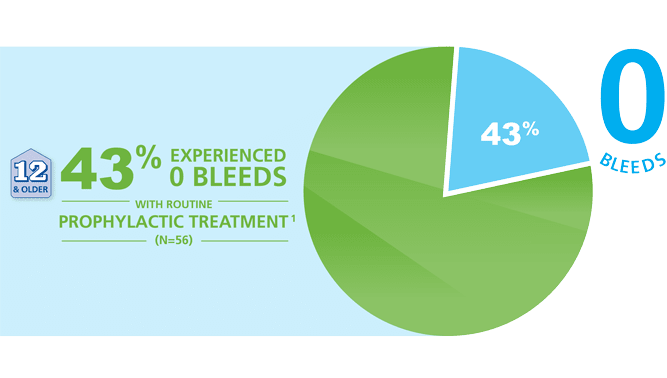
Prophylactic efficacy of RIXUBIS was studied in 56 previously treated patients (PTPs) between 12 and 65 years of age with severe or moderately severe hemophilia B for a mean treatment duration of 6 months.6
Routine prophylactic treatment helped patients achieve a low Annualized Bleed Rate (ABR)*1
(range: 0.0-23.4)
Median ABR (range: 0.0-15.6)
Median ABR (range: 0.0-21.5)
*The majority of patients ≥12 years taking RIXUBIS had joint disease (88%) and target joints (66%) at screening.
Many patients (24 out of 56) taking RIXUBIS® did not bleed over the course of the clinical study1
- In this study, many patients experienced 0 bleeds with RIXUBIS twice-weekly for a mean treatment duration of 6.2 months.6
Most bleeds (211 out of 249) were resolved with 1 to 2 infusions1

With RIXUBIS, 96% of bleeding episodes were rated as having excellent or good bleed resolution.*1
*Excellent is defined as full relief of pain and cessation of objective signs of bleeding after a single infusion; no additional infusion is required for the control of bleeding; Good is defined as definite pain relief and/or improvement in signs of bleeding after a single infusion; possibly requires more than one infusion for complete resolution.
Selected Important Risk Information for RIXUBIS [Coagulation Factor IX (Recombinant)]
What are the possible side effects of RIXUBIS?
Allergic reactions have been reported with RIXUBIS. Call your healthcare provider or get emergency treatment right away if you get a rash or hives, itching, tightness of the throat, chest pain or tightness, difficulty breathing, lightheadedness, dizziness, nausea, or fainting.
Some common side effects of RIXUBIS were unusual taste in the mouth and limb pain.
Tell your healthcare provider about any side effects that bother you or do not go away.
The clinical trial demonstrated that recovery levels were consistent over time at week 261

Recovery levels
WHAT DOES RECOVERY MEAN?
Recovery is the name for the relative amount of factor IX in the bloodstream immediately after infusing. An average recovery is about 50%.3 Factor levels gradually decrease following infusion. Recovery is different for every person and may change for you over time. It can be influenced by age and weight and differs based on the factor product used.4,5
Talk to your doctor about what recovery levels mean for you.
![RIXUBIS [Coagulation Factor IX (Recombinant)] Logo.](/dist/images/Rixubis-header-logo.png)

